|
By obtaining an electron, halogens form anions with a -1 charge. They have 7 electrons in their outermost shell and desire to gain an extra electron to complete their shell of 8 electrons. The halogens are so reactive due to their electronic configuration. Halogens are the most reactive nonmetals on the periodic table. Halogens, unlike metals, exist in all three different states of matter in their standard state- fluorine is found naturally as a gas, bromine as a liquid, and the larger iodine is found naturally as a solid. This group includes fluorine, chlorine, bromine, iodine, and the more recently discover astatine. Group 17 can be found in the table’s 17th column. The halogens make up Group 17 of the elements on the periodic table. The name halogen means “salt-producer.” They are Halogens make up group 17, which is found in the 17th column of the periodic table. Where are the halogens found on the periodic table? 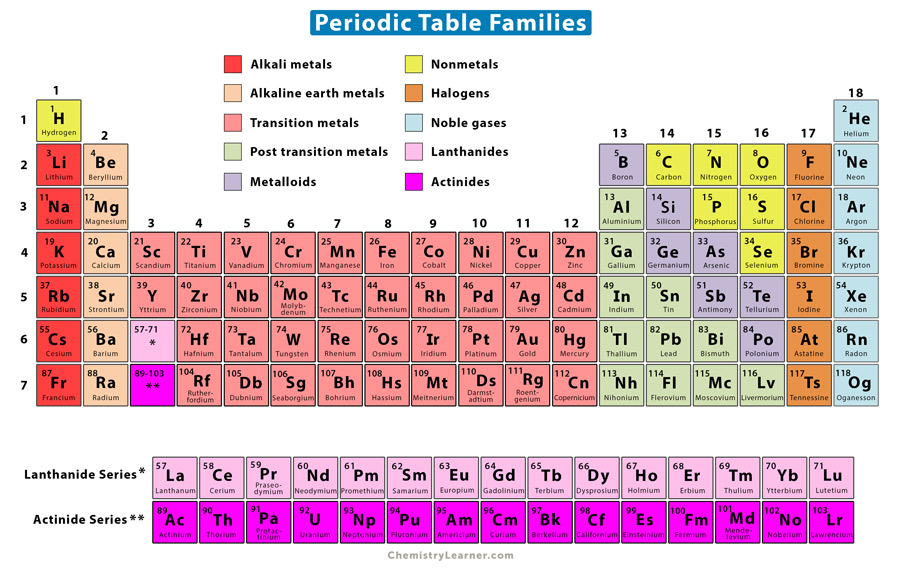
This makes them very reactive as they seek to complete their outermost shell of 8 electrons.ģ. How many valence electrons do halogens have? Their name means salt-producer, and they are the most reactive non-metal elements.Ģ. The halogens are a group of elements found in group 17 of the periodic table. Elements in the periodic table can be described as metals, metalloids, and nonmetals. 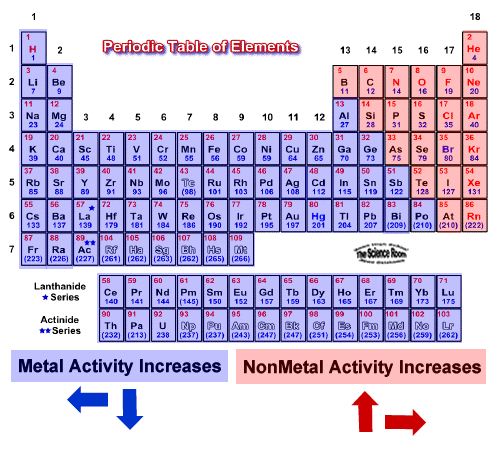 
Elements in the same group typically have similar chemical properties as a result of their similar electronic configuration. All of the elements in a group share the same number of valence electrons: electrons in their outermost shell. Refresher: The periodic table is organized into groups - where each column comprises a group.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
